
荧光标记或荧光探针是众多成像和诊断应用的理想选择。荧光多肽的应用范围包括肽与蛋白质相互作用研究、酶活性测定或新疾病模型的开发。荧光团可以共价连接到N端或C端,但建议与N端连接。由受体和猝灭剂组成的FRET对可以在内部或外部连接。对于较长的序列,建议在内部连接FRET对。
黄金城集团提供了大量的多肽荧光标记,且种类还在不断增加。下方列出了一些黄金城集团最常用的修饰:
| 名称 | 激发波长(nm) | 发射波长(nm) | 发射颜色 | 应用领域 |
|---|---|---|---|---|
| 7-甲氧基香豆素-4-乙酸 | 328 | 393 | 蓝色 |
体外成像 亚细胞定位 共聚焦显微镜 流式细胞术 |
| FITC-Ahx | 494 | 521 | 绿色 | |
| FAM | 495 | 520 | 绿色 | |
| Cy3 | 555 | 570 | 黄色 | |
| 5-羧基四甲基罗丹明(TMR) | 542 | 568 | 橙色 | |
| Cy5 | 646 | 662 | 红色 | |
| Cy5.5 | 673 | 707 | 近红外 |
体外成像 亚细胞定位 体内光学成像 血管造影术 |
| Cy7 | 750 | 773 | 近红外 |
荧光共振能量转移(FRET)是一种描述两种荧光团之间能量转移的机制。由于FRET效率部分取决于供体分子和受体分子之间的距离,因此该技术通常用于研究酶的效率、蛋白质与蛋白质之间相互作用或其他分子动力学(图1)。

图1. 用于蛋白酶研究的FRET机制。当肽保持完整时,受体分子将淬灭供体分子,检测不到荧光。如果肽序列被蛋白酶活性裂解,受体将不再淬灭供体,从而检测到荧光信号。
| 供体 | 受体 | ||||
|---|---|---|---|---|---|
| 名称 | 激发波长(nm) | 发射波长(nm) | 名称 | 激发波长(nm) | 发射波长(nm) |
| Cy2 | 490 | 510 | Cy3 | 555 | 570 |
| FITC | 494 | 521 | TRITC | 557 | 576 |
| FAM | 495 | 520 | Cy3 | 555 | 570 |
| FAM | 495 | 520 | 德克萨斯红 | 589 | 615 |
| FAM | 495 | 520 | Cy5 | 646 | 662 |
| Cy3 | 555 | 570 | Cy5 | 646 | 662 |
| EDANS | 335 | 493 | DABCYL | 453 | - |
| Glu(EDANS)-NH2 | 335 | 493 | DABCYL | 453 | - |
| MCA | 328 | 393 | DNP | 348 | - |
| Abz | 330 | 420 | DNP | 348 | - |
| Abz | 330 | 420 | Tyr (3-NO2) | 360 | - |
利用共焦显微镜或荧光显微镜进行体外成像仍然是研究细胞内各种生物学过程和相互作用的最高效、最有效的方法之一。当与成像技术结合时,荧光标记多肽可用于识别特定靶标。 阅读更多 »
For example, cell penetrating peptides (CPPs) modified with FITC and photoactivatable probes have been used to track binding patterns and dynamic behavior over time. Unlike proteins, these peptides localize to specific targets on actin and are less prone to protein aggregation, making them ideal for in vitro tracking (Pan 2014). Similarly, FITC-labeled CPPs have been used to image intracellular compartments of cells with low risk of cytotoxicity (Kirkham 2015).
用于血管造影的体内成像仍然是荧光标记多肽的主要应用领域。该技术使用造影剂对血管内部进行成像,使医生能够为个人选择最合适的医疗策略或治疗方法。 阅读更多 »
For example, high resolution near-infrared fluorophores have been used as fibrin imaging-agents for deep vein thrombosis (DVT). Using Cy7 labeled fibrin-targeting peptides, CT scanning and confocal microscopy were used to differentiate between acute and subacute murine DVT (Hara 2012). Similar techniques have also been used for detecting apoptosis as a symptom for glaucoma. Fluorophore-labeled peptides that are activated by caspases can be imaged in vivo to track glaucoma progression (Qiu 2014).
蛋白水解酶在传染性疾病中发挥重要作用,使其成为开发新疗法的研究目标。为了识别这些酶靶向的肽序列,通常使用肽文库。潜在的蛋白酶靶序列与FRET对结合在一起,这样当酶裂解靶肽时,可以检测到荧光信号。 阅读更多 »
In these studies, a donor molecule, such as Abz (Marcondes 2015) or Lucifer Yellow (Rossé 2000) is covalently attached to the C-terminus, while acceptor molecules (ex. Dabsyl, DNP) are coupled to the N-terminus. This strategy is commonly employed with peptides synthesized using a solid-phase approach, and peptides are left conjugated to the bead. If the peptides are not targeted by protease activity, both the donor and acceptor will be present, and the bead will appear non-fluorescent. If the peptides are cleaved, the peptide will no longer be quenched by FRET and the beads will fluoresce. Since protease studies are most effective when FRET is combined with shorter peptides, micro-scale peptide libraries are ideal choices for these experiments.
荧光多肽标记有许多研究应用,黄金城集团在合成具有各种修饰的肽方面拥有丰富的经验。
案例研究1
序列:LYRLGLGH
修饰:MCA/DNP
数量:1-4 mg
| 要求纯度 | 预计交付时间 | 实际纯度 | 实际交付时间 |
|---|---|---|---|
>98%
|
17天 |
99.563%
|
14天 |
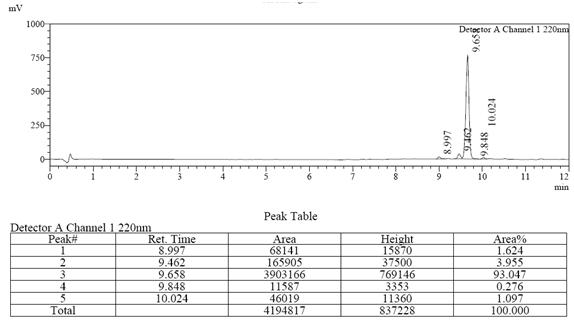
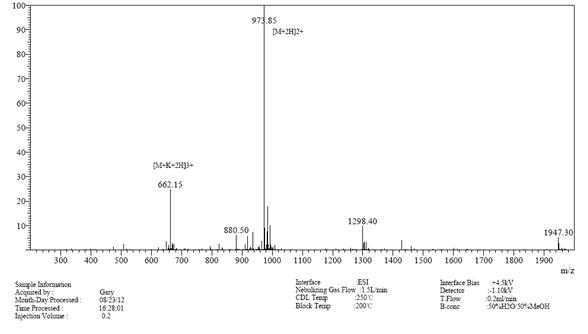
案例研究2
序列:IKDLSKEERLWEVQRILTALKRKLREA
修饰:5-FAM (N端)
数量:10-14 mg
| 要求纯度 | 预计交付时间 | 实际纯度 | 实际交付时间 |
|---|---|---|---|
| >98% | 23天 | 99.10% | 13天 |
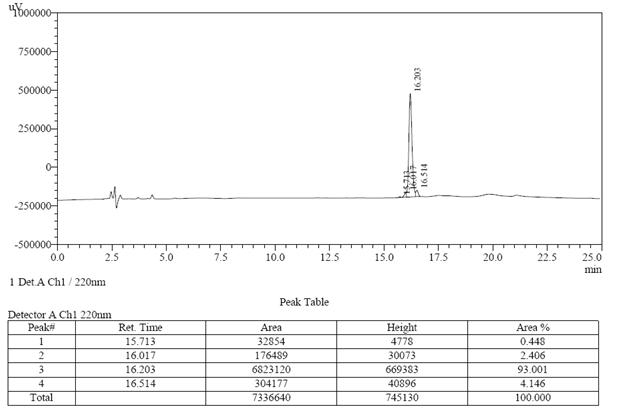
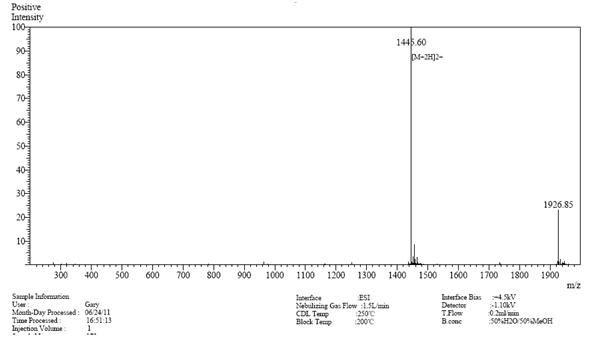
案例研究3
序列:RAKWNNTLKQIASK
修饰: FITC-Ahx (N端)
数量: 5-9 mg
| 要求纯度 | 预计交付时间 | 实际纯度 | 实际交付时间 |
|---|---|---|---|
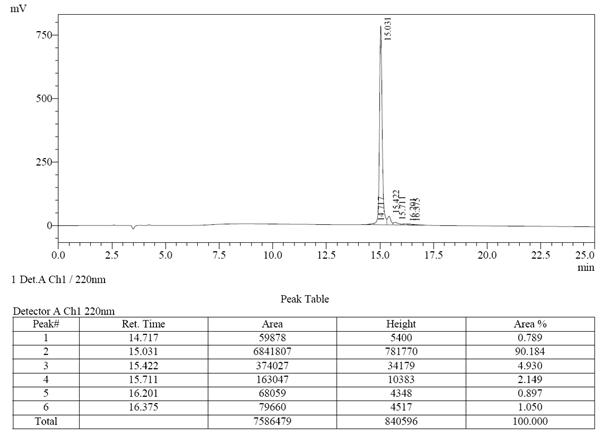
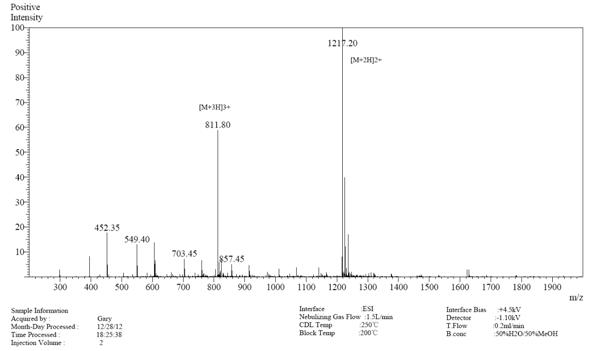
| >98% | 17天 | 99.81% | 5天 |

<tr id='0roef'><strong id='0roef'></strong><small id='0roef'></small><button id='0roef'></button><li id='0roef'><noscript id='0roef'><big id='0roef'></big><dt id='0roef'></dt></noscript></li></tr><ol id='0roef'><option id='0roef'><table id='0roef'><blockquote id='0roef'><tbody id='0roef'></tbody></blockquote></table></option></ol><u id='0roef'></u><kbd id='0roef'><kbd id='0roef'></kbd></kbd>
